Original: $54.00
-70%$54.00
$16.20The Story
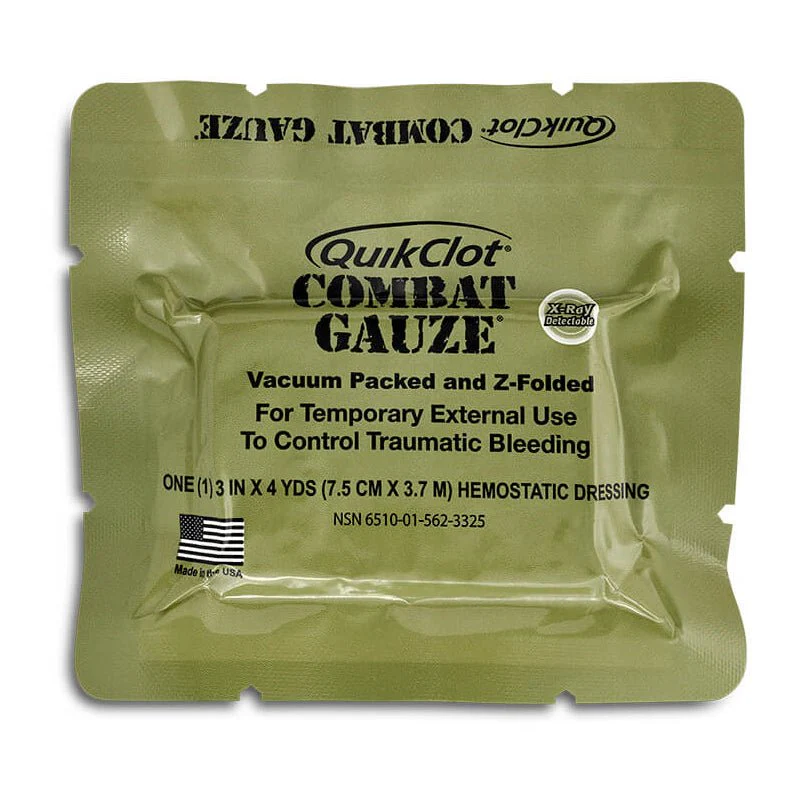
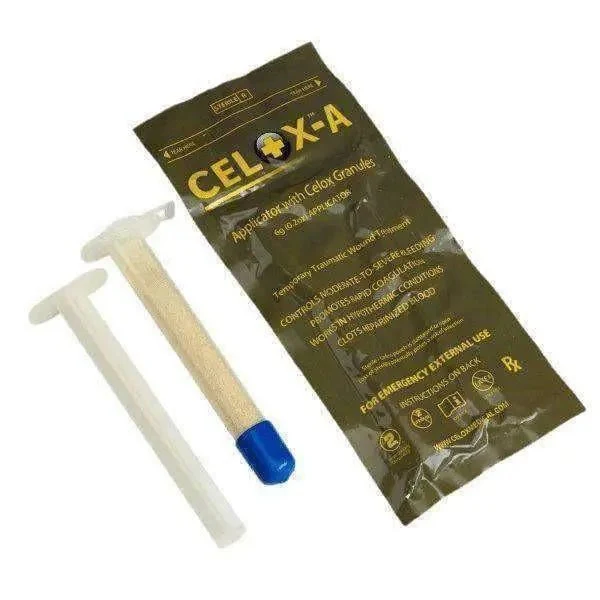
QuikClot Combat Gauze by Z-Medica (Teleflex) is the CoTCCC-recommended hemostatic dressing of choice for the U.S. Department of Defense — the primary first-line hemostatic agent used at point of injury by all branches of the U.S. Military. It is a sterile, kaolin-impregnated Z-fold gauze (3 in. × 4 yds.) that activates Factor XII to accelerate the body's clotting cascade without generating heat, packaged in a vacuum-sealed, 8-tear-point foil pouch.
Key Specifications
| Specification | Detail |
|---|---|
| Manufacturer | Z-Medica, LLC (a Teleflex company) |
| SKU / MED-TAC Item # | MEDTAC0491 |
| NSN | 6510-01-562-3325 |
| Active Ingredient | Kaolin (inorganic mineral — no animal or human proteins) |
| Gauze Dimensions | 3 in. × 4 yds. (7.5 cm × 3.7 m) — Z-fold |
| Packaged Dimensions | 6 in. H × 4 in. W × 0.6 in. D |
| Weight (packaged) | ~0.8 oz (23 g) |
| Format | Z-fold, vacuum-sealed military-grade foil pouch, 8 tear points |
| X-Ray Detectable | Yes — radiopaque strip included |
| Shelf Life | 5 years from date of manufacture |
| CoTCCC Status | Recommended — Primary hemostatic dressing of choice (since 2008) |
| DAPA Listed | Yes |
| Origin | Made in the USA |
Product Overview
Since April 2008, QuikClot Combat Gauze has been the CoTCCC-recommended hemostatic dressing of choice for the U.S. Department of Defense — a distinction it has held continuously for over 15 years. It is the only hemostatic gauze carried by all branches of the U.S. Military for point-of-injury control of life-threatening hemorrhage. The product is manufactured by Z-Medica (a Teleflex company) in the United States and impregnated with kaolin, an inorganic clay mineral that activates Factor XII in the coagulation cascade, accelerating the body's natural clotting response without generating heat and without containing animal or human-derived proteins.
The mechanism of action distinguishes Combat Gauze from chitosan-based competitors: kaolin activates the intrinsic clotting pathway at the Factor XII (Hageman factor) level, initiating a cascade that produces thrombin and ultimately cross-linked fibrin — a structurally robust clot. This approach works synergistically with the body's own hemostatic system and has been validated in peer-reviewed literature. A 2011 study published in the Journal of Trauma (Kheirabadi et al.) demonstrated superior performance for kaolin-based gauze in a swine junctional hemorrhage model compared to plain gauze, with significantly reduced blood loss and improved survival. The U.S. Naval Medical Research Center and the U.S. Army Institute of Surgical Research independently validated Combat Gauze's efficacy, providing the evidence base for the original CoTCCC recommendation in 2008.
The Z-fold configuration allows one-handed deployment and rapid wound packing under stress. The vacuum-sealed, military-grade foil pouch features eight tear points — allowing access from any corner — and compresses to a compact cube (6 × 4 × 0.6 in.) that fits standard IFAK and blowout kit pouches. An embedded radiopaque strip ensures the dressing can be identified on X-ray imaging, a critical safety feature for surgical teams receiving pre-packed wounds. The 5-year shelf life and ambient temperature storage (no refrigeration required) make it ideal for pre-positioned medical kits in vehicles, buildings, and military contexts. Storage should avoid direct sunlight and prolonged high heat, but no special cold-chain requirements apply.
How to Apply
- Expose the wound and identify the source of bleeding.
- Tear open the foil pouch at any of the 8 tear points and remove the Z-folded gauze.
- Place one end of the gauze directly on the bleeding source and pack tightly into the wound cavity, following the wound track.
- Continue packing, feeding in the unfolding gauze, until the wound is filled above skin level.
- Apply firm, direct pressure for a minimum of 3 minutes (5 minutes recommended for arterial bleeding). Do not lift to inspect — maintain continuous pressure.
- Secure with a pressure bandage and monitor for re-bleeding during transport.
- Mark time of application on the patient and report to receiving medical personnel.
Comparison Context
QuikClot Combat Gauze is the benchmark CoTCCC-recommended hemostatic gauze against which all alternatives are measured. Chitosan-based alternatives — including Celox Rapid, ChitoGauze XR PRO, and ChitoSAM 100 — work via a clotting-cascade-independent mechanism (chitosan muco-adhesion to red blood cell membranes) and were added to the CoTCCC guidelines in 2014 as acceptable alternatives, particularly for patients on anticoagulant therapy or with hypothermia-induced coagulopathy where the intrinsic clotting cascade may be impaired. Celox products require only 60 seconds of compression (vs. 3 minutes for Combat Gauze) in laboratory testing, though operational data from DoD theater use continues to support Combat Gauze as the primary choice. Combat Gauze does not generate heat — an important distinction from older zeolite-based QuikClot powder products that are no longer in production.
For complete massive hemorrhage control kits that pair Combat Gauze with a tourniquet and pressure bandage, see MED-TAC's full hemostatic agents collection or IFAK & First Aid Kits.
All products sourced from the actual brand manufacturer or authorized master distributors. CoTCCC recommendation status verified where applicable. Ships from MED-TAC International, Pembroke Pines, FL — clinician-founded, veteran-led, SDVOSB-certified.

Details & Craftsmanship
Every detail has been carefully considered to bring you the perfect product.

Details & Craftsmanship
Every detail has been carefully considered to bring you the perfect product.
Details & Craftsmanship
Every detail has been carefully considered to bring you the perfect product.
Description
QuikClot Combat Gauze by Z-Medica (Teleflex) is the CoTCCC-recommended hemostatic dressing of choice for the U.S. Department of Defense — the primary first-line hemostatic agent used at point of injury by all branches of the U.S. Military. It is a sterile, kaolin-impregnated Z-fold gauze (3 in. × 4 yds.) that activates Factor XII to accelerate the body's clotting cascade without generating heat, packaged in a vacuum-sealed, 8-tear-point foil pouch.
Key Specifications
| Specification | Detail |
|---|---|
| Manufacturer | Z-Medica, LLC (a Teleflex company) |
| SKU / MED-TAC Item # | MEDTAC0491 |
| NSN | 6510-01-562-3325 |
| Active Ingredient | Kaolin (inorganic mineral — no animal or human proteins) |
| Gauze Dimensions | 3 in. × 4 yds. (7.5 cm × 3.7 m) — Z-fold |
| Packaged Dimensions | 6 in. H × 4 in. W × 0.6 in. D |
| Weight (packaged) | ~0.8 oz (23 g) |
| Format | Z-fold, vacuum-sealed military-grade foil pouch, 8 tear points |
| X-Ray Detectable | Yes — radiopaque strip included |
| Shelf Life | 5 years from date of manufacture |
| CoTCCC Status | Recommended — Primary hemostatic dressing of choice (since 2008) |
| DAPA Listed | Yes |
| Origin | Made in the USA |
Product Overview
Since April 2008, QuikClot Combat Gauze has been the CoTCCC-recommended hemostatic dressing of choice for the U.S. Department of Defense — a distinction it has held continuously for over 15 years. It is the only hemostatic gauze carried by all branches of the U.S. Military for point-of-injury control of life-threatening hemorrhage. The product is manufactured by Z-Medica (a Teleflex company) in the United States and impregnated with kaolin, an inorganic clay mineral that activates Factor XII in the coagulation cascade, accelerating the body's natural clotting response without generating heat and without containing animal or human-derived proteins.
The mechanism of action distinguishes Combat Gauze from chitosan-based competitors: kaolin activates the intrinsic clotting pathway at the Factor XII (Hageman factor) level, initiating a cascade that produces thrombin and ultimately cross-linked fibrin — a structurally robust clot. This approach works synergistically with the body's own hemostatic system and has been validated in peer-reviewed literature. A 2011 study published in the Journal of Trauma (Kheirabadi et al.) demonstrated superior performance for kaolin-based gauze in a swine junctional hemorrhage model compared to plain gauze, with significantly reduced blood loss and improved survival. The U.S. Naval Medical Research Center and the U.S. Army Institute of Surgical Research independently validated Combat Gauze's efficacy, providing the evidence base for the original CoTCCC recommendation in 2008.
The Z-fold configuration allows one-handed deployment and rapid wound packing under stress. The vacuum-sealed, military-grade foil pouch features eight tear points — allowing access from any corner — and compresses to a compact cube (6 × 4 × 0.6 in.) that fits standard IFAK and blowout kit pouches. An embedded radiopaque strip ensures the dressing can be identified on X-ray imaging, a critical safety feature for surgical teams receiving pre-packed wounds. The 5-year shelf life and ambient temperature storage (no refrigeration required) make it ideal for pre-positioned medical kits in vehicles, buildings, and military contexts. Storage should avoid direct sunlight and prolonged high heat, but no special cold-chain requirements apply.
How to Apply
- Expose the wound and identify the source of bleeding.
- Tear open the foil pouch at any of the 8 tear points and remove the Z-folded gauze.
- Place one end of the gauze directly on the bleeding source and pack tightly into the wound cavity, following the wound track.
- Continue packing, feeding in the unfolding gauze, until the wound is filled above skin level.
- Apply firm, direct pressure for a minimum of 3 minutes (5 minutes recommended for arterial bleeding). Do not lift to inspect — maintain continuous pressure.
- Secure with a pressure bandage and monitor for re-bleeding during transport.
- Mark time of application on the patient and report to receiving medical personnel.
Comparison Context
QuikClot Combat Gauze is the benchmark CoTCCC-recommended hemostatic gauze against which all alternatives are measured. Chitosan-based alternatives — including Celox Rapid, ChitoGauze XR PRO, and ChitoSAM 100 — work via a clotting-cascade-independent mechanism (chitosan muco-adhesion to red blood cell membranes) and were added to the CoTCCC guidelines in 2014 as acceptable alternatives, particularly for patients on anticoagulant therapy or with hypothermia-induced coagulopathy where the intrinsic clotting cascade may be impaired. Celox products require only 60 seconds of compression (vs. 3 minutes for Combat Gauze) in laboratory testing, though operational data from DoD theater use continues to support Combat Gauze as the primary choice. Combat Gauze does not generate heat — an important distinction from older zeolite-based QuikClot powder products that are no longer in production.
For complete massive hemorrhage control kits that pair Combat Gauze with a tourniquet and pressure bandage, see MED-TAC's full hemostatic agents collection or IFAK & First Aid Kits.
All products sourced from the actual brand manufacturer or authorized master distributors. CoTCCC recommendation status verified where applicable. Ships from MED-TAC International, Pembroke Pines, FL — clinician-founded, veteran-led, SDVOSB-certified.