The Story
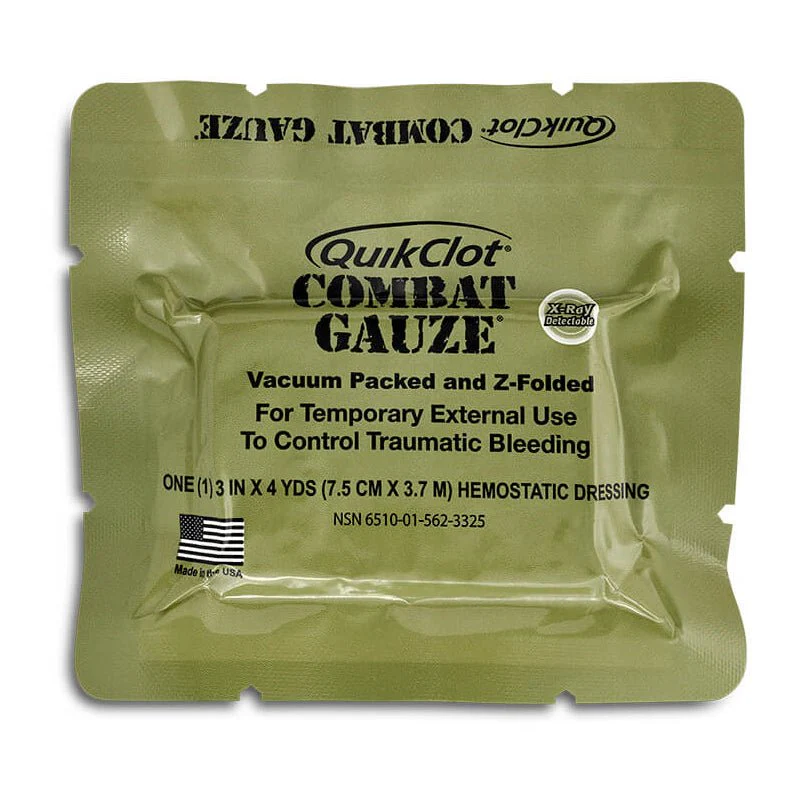
ChitoGAUZE XR PRO by Tricol Biomedical (HemCon) is a CoTCCC-recommended chitosan hemostatic gauze — a sterile, 3 in. × 4 yd. Z-fold dressing coated with Chito+ chitosan that controls severe arterial bleeding independently of the clotting cascade. It features a single embedded X-ray detectable thread, vacuum-sealed packaging (5.75 × 5.0 × 0.65 in.), antimicrobial activity against gram-positive and gram-negative bacteria, and up to 48-hour wound dwell time. MED-TAC SKU: MEDTAC0628.
Key Specifications
| Specification | Detail |
|---|---|
| Manufacturer | Tricol Biomedical / HemCon Medical Technologies (Portland, OR) |
| SKU / MED-TAC Item # | MEDTAC0628 | Tricol Item #1090 |
| Active Ingredient | Chitosan (Chito+ technology) — uniform coating on polyester/rayon blend gauze |
| Gauze Dimensions | 3 in. × 4 yds. (7.5 cm × 3.7 m) — Z-fold |
| Packaged Dimensions | 5.75 in. H × 5.0 in. W × 0.65 in. D |
| X-Ray Detectable Strips | Single embedded radiopaque thread (XR PRO) |
| Format | Z-fold, vacuum-sealed foil pouch |
| Antimicrobial | Broad spectrum — gram-positive and gram-negative organisms including MRSA |
| CoTCCC Status | Recommended — chitosan-based alternative (TCCC Guideline Change 13-05, 2014) |
| Dwell Time | Up to 48 hours |
| Latex Free | Yes |
| Shelf Life | 5 years |
Product Overview
ChitoGAUZE XR PRO is the foundational product in HemCon's CoTCCC-recommended chitosan gauze line. It is composed of a polyester/rayon blend non-woven medical gauze uniformly coated with Chito+ chitosan — a proprietary formulation engineered for maximum chitosan-blood interaction and optimal wound conformance. The gauze is Z-folded to a compact packaged size and vacuum-sealed in a rugged foil pouch designed for battlefield and austere environment durability. A single embedded radiopaque filament (the "XR" designation) allows X-ray detection for imaging verification at definitive care.
The Chito+ mechanism operates via electrostatic muco-adhesion — the positively charged chitosan molecules bind directly to the negatively charged membranes of red blood cells, causing aggregation and physical sealing of the wound. This mechanism is entirely independent of the enzymatic coagulation cascade, distinguishing it from kaolin-based hemostatics (QuikClot Combat Gauze) that rely on Factor XII activation. The clinical implication is significant: in patients with impaired clotting cascade function — hypothermic patients, those on anticoagulant therapy, or those with trauma-induced coagulopathy — chitosan-based hemostatics like ChitoGAUZE XR PRO maintain efficacy where kaolin-based products may be less effective.
Beyond hemostasis, ChitoGAUZE XR PRO's chitosan coating provides inherent antimicrobial activity against a broad spectrum of bacteria — including the clinically critical organisms MRSA, Enterococcus faecalis, and Acinetobacter baumannii. This is particularly relevant in combat wound management, where contamination is common and antibiotic treatment may be delayed. The 48-hour maximum dwell time provides flexibility for prolonged field care scenarios and evacuation timelines. The gauze is removed by gently peeling from the wound surface — the non-shedding design ensures no chitosan fragments remain in the wound. ChitoGAUZE XR PRO is FDA-cleared for external, temporary control of severely bleeding wounds and is gamma-irradiated for sterility.
How to Apply
- Identify and expose the source of bleeding.
- Open the vacuum-sealed pouch and remove the Z-fold gauze.
- Place the dressing directly on the source of bleeding and apply direct digital pressure.
- Pack completely into the wound track, ensuring contact with all bleeding surfaces.
- Apply firm, sustained pressure for a minimum of 3 minutes. Do not lift to inspect.
- If trimming the dressing, ensure the radiopaque element remains in the portion applied to the wound.
- Secure with a pressure bandage. May remain in place up to 48 hours; remove at definitive care.
XR PRO vs. XR2 PRO vs. Combat Gauze
ChitoGAUZE XR PRO and XR2 PRO have identical hemostatic performance — the only difference is the XR2's additional (second) X-ray detectable strip for enhanced imaging reliability at hospital handoff. For field use, both are equivalent; for units where surgical team imaging requirements mandate dual-strip gauze, XR2 PRO is preferred. Both products offer the same advantages over QuikClot Combat Gauze: independent of clotting cascade, antimicrobial properties, and 48-hour dwell time.
Complete your hemorrhage control system with MED-TAC's full hemostatic agents collection, massive hemorrhage control kits, and IFAK kits.
All products sourced from the actual brand manufacturer or authorized master distributors. CoTCCC recommendation status verified where applicable. Ships from MED-TAC International, Pembroke Pines, FL — clinician-founded, veteran-led, SDVOSB-certified.
Details & Craftsmanship
Every detail has been carefully considered to bring you the perfect product.
Details & Craftsmanship
Every detail has been carefully considered to bring you the perfect product.
Description
ChitoGAUZE XR PRO by Tricol Biomedical (HemCon) is a CoTCCC-recommended chitosan hemostatic gauze — a sterile, 3 in. × 4 yd. Z-fold dressing coated with Chito+ chitosan that controls severe arterial bleeding independently of the clotting cascade. It features a single embedded X-ray detectable thread, vacuum-sealed packaging (5.75 × 5.0 × 0.65 in.), antimicrobial activity against gram-positive and gram-negative bacteria, and up to 48-hour wound dwell time. MED-TAC SKU: MEDTAC0628.
Key Specifications
| Specification | Detail |
|---|---|
| Manufacturer | Tricol Biomedical / HemCon Medical Technologies (Portland, OR) |
| SKU / MED-TAC Item # | MEDTAC0628 | Tricol Item #1090 |
| Active Ingredient | Chitosan (Chito+ technology) — uniform coating on polyester/rayon blend gauze |
| Gauze Dimensions | 3 in. × 4 yds. (7.5 cm × 3.7 m) — Z-fold |
| Packaged Dimensions | 5.75 in. H × 5.0 in. W × 0.65 in. D |
| X-Ray Detectable Strips | Single embedded radiopaque thread (XR PRO) |
| Format | Z-fold, vacuum-sealed foil pouch |
| Antimicrobial | Broad spectrum — gram-positive and gram-negative organisms including MRSA |
| CoTCCC Status | Recommended — chitosan-based alternative (TCCC Guideline Change 13-05, 2014) |
| Dwell Time | Up to 48 hours |
| Latex Free | Yes |
| Shelf Life | 5 years |
Product Overview
ChitoGAUZE XR PRO is the foundational product in HemCon's CoTCCC-recommended chitosan gauze line. It is composed of a polyester/rayon blend non-woven medical gauze uniformly coated with Chito+ chitosan — a proprietary formulation engineered for maximum chitosan-blood interaction and optimal wound conformance. The gauze is Z-folded to a compact packaged size and vacuum-sealed in a rugged foil pouch designed for battlefield and austere environment durability. A single embedded radiopaque filament (the "XR" designation) allows X-ray detection for imaging verification at definitive care.
The Chito+ mechanism operates via electrostatic muco-adhesion — the positively charged chitosan molecules bind directly to the negatively charged membranes of red blood cells, causing aggregation and physical sealing of the wound. This mechanism is entirely independent of the enzymatic coagulation cascade, distinguishing it from kaolin-based hemostatics (QuikClot Combat Gauze) that rely on Factor XII activation. The clinical implication is significant: in patients with impaired clotting cascade function — hypothermic patients, those on anticoagulant therapy, or those with trauma-induced coagulopathy — chitosan-based hemostatics like ChitoGAUZE XR PRO maintain efficacy where kaolin-based products may be less effective.
Beyond hemostasis, ChitoGAUZE XR PRO's chitosan coating provides inherent antimicrobial activity against a broad spectrum of bacteria — including the clinically critical organisms MRSA, Enterococcus faecalis, and Acinetobacter baumannii. This is particularly relevant in combat wound management, where contamination is common and antibiotic treatment may be delayed. The 48-hour maximum dwell time provides flexibility for prolonged field care scenarios and evacuation timelines. The gauze is removed by gently peeling from the wound surface — the non-shedding design ensures no chitosan fragments remain in the wound. ChitoGAUZE XR PRO is FDA-cleared for external, temporary control of severely bleeding wounds and is gamma-irradiated for sterility.
How to Apply
- Identify and expose the source of bleeding.
- Open the vacuum-sealed pouch and remove the Z-fold gauze.
- Place the dressing directly on the source of bleeding and apply direct digital pressure.
- Pack completely into the wound track, ensuring contact with all bleeding surfaces.
- Apply firm, sustained pressure for a minimum of 3 minutes. Do not lift to inspect.
- If trimming the dressing, ensure the radiopaque element remains in the portion applied to the wound.
- Secure with a pressure bandage. May remain in place up to 48 hours; remove at definitive care.
XR PRO vs. XR2 PRO vs. Combat Gauze
ChitoGAUZE XR PRO and XR2 PRO have identical hemostatic performance — the only difference is the XR2's additional (second) X-ray detectable strip for enhanced imaging reliability at hospital handoff. For field use, both are equivalent; for units where surgical team imaging requirements mandate dual-strip gauze, XR2 PRO is preferred. Both products offer the same advantages over QuikClot Combat Gauze: independent of clotting cascade, antimicrobial properties, and 48-hour dwell time.
Complete your hemorrhage control system with MED-TAC's full hemostatic agents collection, massive hemorrhage control kits, and IFAK kits.
All products sourced from the actual brand manufacturer or authorized master distributors. CoTCCC recommendation status verified where applicable. Ships from MED-TAC International, Pembroke Pines, FL — clinician-founded, veteran-led, SDVOSB-certified.